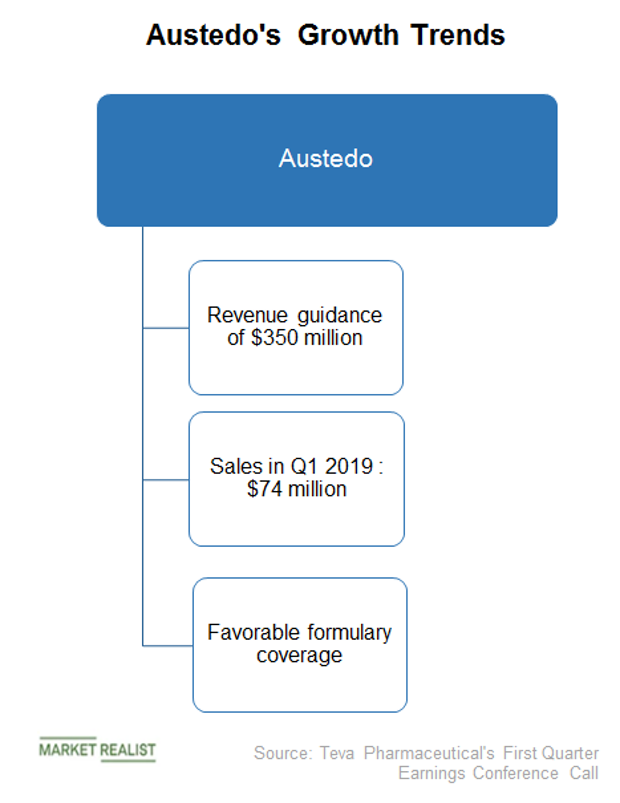
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena

Teva Announces Post Hoc Analysis of Long-Term Data Examining Treatment with AUSTEDO® (deutetrabenazine) Tablets in Adult Patients with Tardive Dyskinesia

Austedo for Huntington's disease: Teva Pharmaceuticals has found a nifty way to keep drugs in your body for longer — Quartz

Teva's Austedo (deutetrabenazine) Receives the NMPA's Approval to Treat Chorea Associated with Huntington's Disease and Tardive Dyskinesia in Adults

Punit on Twitter: "#Austedo (deutetrabenazine) revenue & TRx analysis Q2-17- $1 M Q2-18- $44 M Q2-19- $96 M Q1-20- $122 M Now, Austedo approval in China may increase #API demand. Also,dose increases